Citation
Daniel K. Nielsen, Laura L. Nielsen, Spencer B. Jones, Lawrence Toll, Matthew C. Asplund, and Steven L. Castle The Journal of Organic Chemistry 2009 74 (3), 1187-1199 DOI: 10.1021/jo802370v
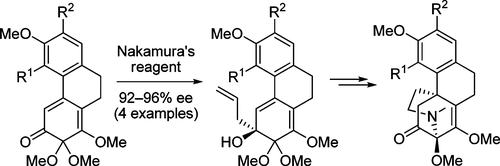
A synthesis of the hasubanan alkaloids hasubanonine, runanine, and aknadinine via a unified route was attempted. Construction of key phenanthrene intermediates by a Suzuki coupling−Wittig olefination−ring-closing metathesis sequence allowed a convergent and flexible approach. Conversion of the phenanthrenes into the target structures was projected to involve six steps including phenolic oxidation, ketone allylation, anionic oxy-Cope rearrangement, and acid-promoted cyclization. The final step was thwarted by a pinacol-like rearrangement that delivered the unnatural isohasubanan alkaloid skeleton. The structures of the products were established by exhaustive NMR experiments and confirmed by GIAO 13C NMR calculations of runanine, isorunanine, and three other isomers. These computations revealed some inconsistencies with the benzene solvent correction which suggest that caution should be used in employing this algorithm. The racemic synthesis of isohasubanonine was transformed into an enantioselective synthesis by the discovery that Nakamura’s chiral bisoxazoline-ligated allylzinc reagent mediates the enantioselective allylation of ketone 19 in 93% ee. This method could be extended to three other structurally related ketones (92−96% ee), and the enantioselective syntheses of two other isohasubanan alkaloids, isorunanine and isoaknadinine, were accomplished. Racemic isohasubanonine was found to be an ineffective analgesic agent.